The interplay between vitamin D and viral infections
Rev Med Virol. 2019 Jan 6:e2032. doi: 10.1002/rmv.2032.
Teymoori-Rad M1, Shokri F2, Salimi V1, Marashi SM1.
📄 Download the PDF from Sci-Hub via Vitamin D Life
Table 1

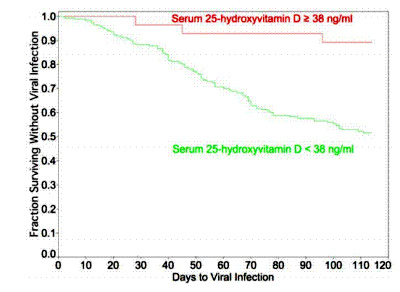
FIGURE 1 The metabolism of vitamin D and its effect on nonimmune and immune cells . (1) Vitamin D is obtained primarily from food, (2) nevertheless sunlight exposure in the skin, where the UV rays alter 7-DHC to pre-vitamin D3, which is the main source of vitamin D3. Previtamin D3 finally is converted into vitamin D3 by heat. (3) all obtained vitamin D is localized to the liver via VDBP. (4) in the liver, 25-hydroxylase converts vitamin D to 25(OH)D. (5) 25(OH) D is transported to kidneys for conversion to 1,25(OH)2D by the action of CYP27B1. (6) 1,25(OH)2D is inactivated and converted to 24,25(OH)2D by CYP24A1. (7) 1,25(OH)2D binds to VDR on target cells. VDR is expressed in various cell jargons such as intestine, bone, kidneys, skin, parathyroid, heart, muscle, eyes, brain, pancreatic beta-cells, and epithelial cells, and also (8) cells of the immune system. (9) on one hand, vitamin D alters some metabolic functions and tissue function (box 1); on the other hand, binding of the metabolites of vitamin D to VDR leads to VDR and RXR heterodimer formation. Consequently, binding of this dimer-to-promoter region of VDREs induces and/or represses transcription of many genes. Expression of VDRs in almost all immune cells indicates that these are one of the main target of vitamin D, and numerous immune biomarkers are modulated via VDRs action (box 2). (10) given the fact that immune cells also express CYP27B1, it can locally facilitate the conversion of 25(OH) D to 1,25(OH)2D. Green color: Induction or increase of expression by vitamin D. red color: Inhibition or decreases of expression by vitamin D. abbreviation: 7-DHC, 7-dihydrocholesterol (7-DHC); 1,25 di-hydroxy-vitamin D [1,25(OH)2D], 25 hydroxy-vitamin D [25(OH)D], cytochrome P450 family 27 subfamily B member 1 (CYP27B1), cytochrome P450 family 24 subfamily a member 1 (CYP24A1), cytochrome P450 family 2 subfamily R member 1 (CYP2R1), retinoic X receptor (RXR), toll-like receptor (TLR), vitamin D receptor (VDR), vitamin D responsive elements (VDRE)
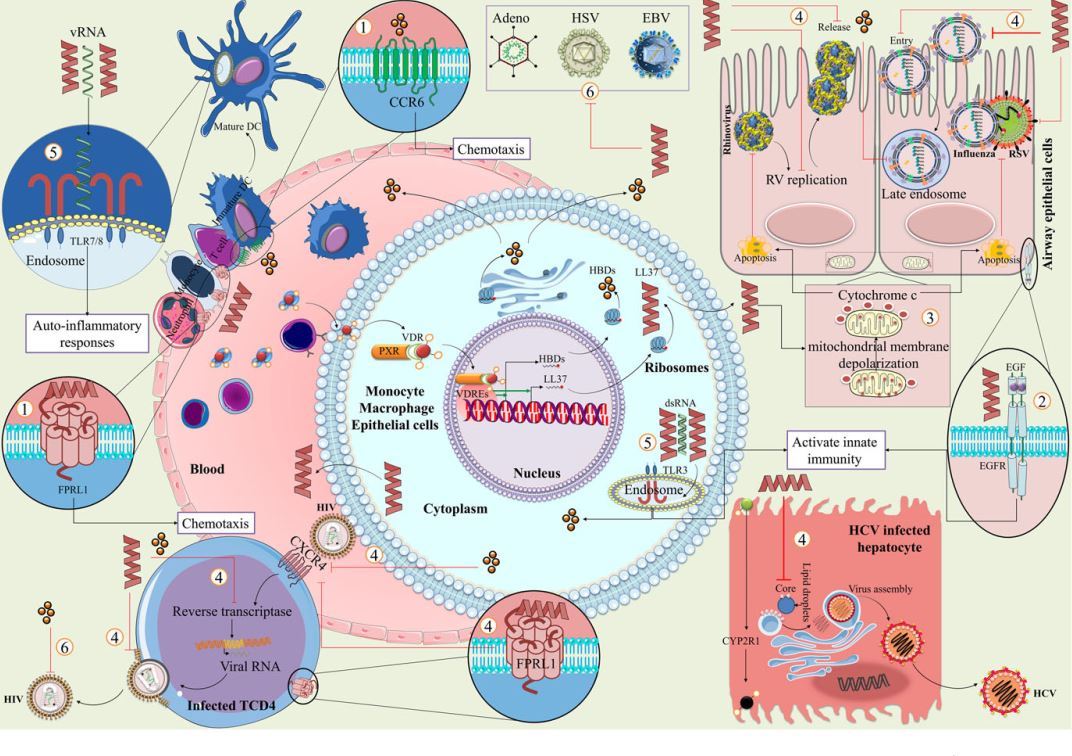
FIGURE 2 Vitamin D and induction of antimicrobial peptides against viral infection . Vitamin D-induced antimicrobial peptides (LL37 and HBDs) via VDR and RXR dimerization act against viral infections through (1) binding to FPRL1 and CCR6 for recruiting immune cells to site of infection; (2) activation of innate immunity by transactivation of the EGFR; (3) clearance of viral infection through mitochondrial membrane depolarization and release of cytochrome c; (4) down-regulation of cell entry, replication, and viral release; (5) protection of viral RNAs from degradation and induced immunity response throughTLRs activation; (6) and direct effects on virions. Abbreviation: Epidermal growth factor receptor (EGFR), formyl peptide receptor-like 1 (FPRL1), human ß-defensins (HBDs), retinoic X receptor (RXR), toll-like receptor (TLR), and vitamin D receptor (VDR)
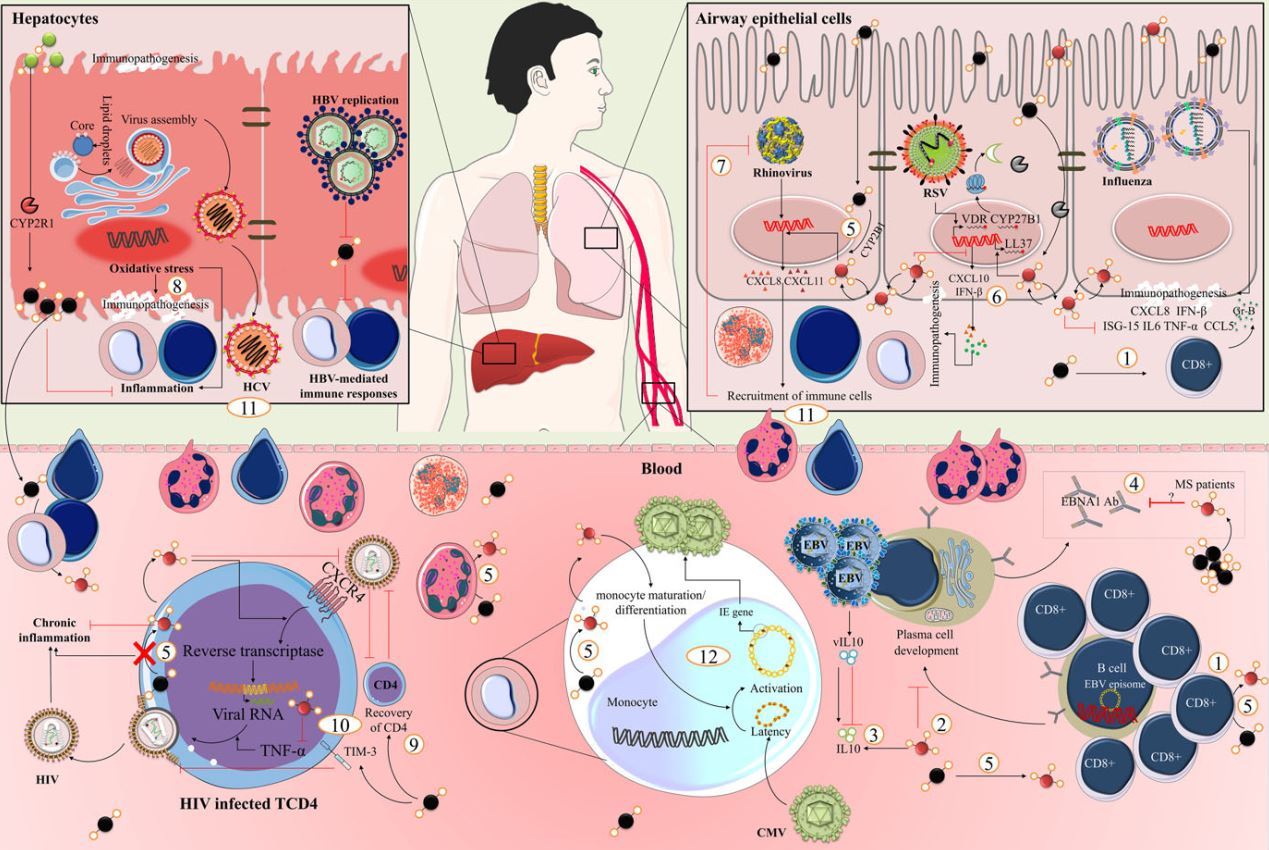
FIGURE 3 Vitamin D and immunoregulatory function . The immunoregulatory functions of vitamin D in viral infections include: (1) increasing virus specific CD8+ T cells (EBV and influenza), (2) inhibition of B cell differentiation to plasma cells and class-switched memory B cells (EBV), (3) compromising the function of IL10-producing regulatory cells (EBV), (4) reduced levels of antibody against viruses available in immune cells (Epstein-Barr nuclear antigen (EBNA)-1), (5) inducing cathelicidin at the site of infection or injury, and therefore synergizing with vitamin D through up-regulation of CYP27B1 by local conversion of more circulating vitamin D to its active form (respiratory viruses), (6) induction of CXCL10 and IFN-p in airway epithelium (RSV), (7) decreased viral replication (HBV and rhinovirus), (8) imbalancing inflammatory process and oxidative stress (HCV), (9) recovery of CD4+ T cells (HIV), (10) facilitating host resistance against viral entry and viral replication (HIV), (11) recruitment of immune cells to the site of infection (respiratory and hepatitis viruses), and (12) alteration of infection phase in latently infected cells (HIV and CMV). Abbreviation: Cytochrome P450 family 27 subfamily B member 1(CYP27B1), IFN-stimulated gene-15 (ISG15), granzyme B (gr-B), vitamin D receptor (VDR)
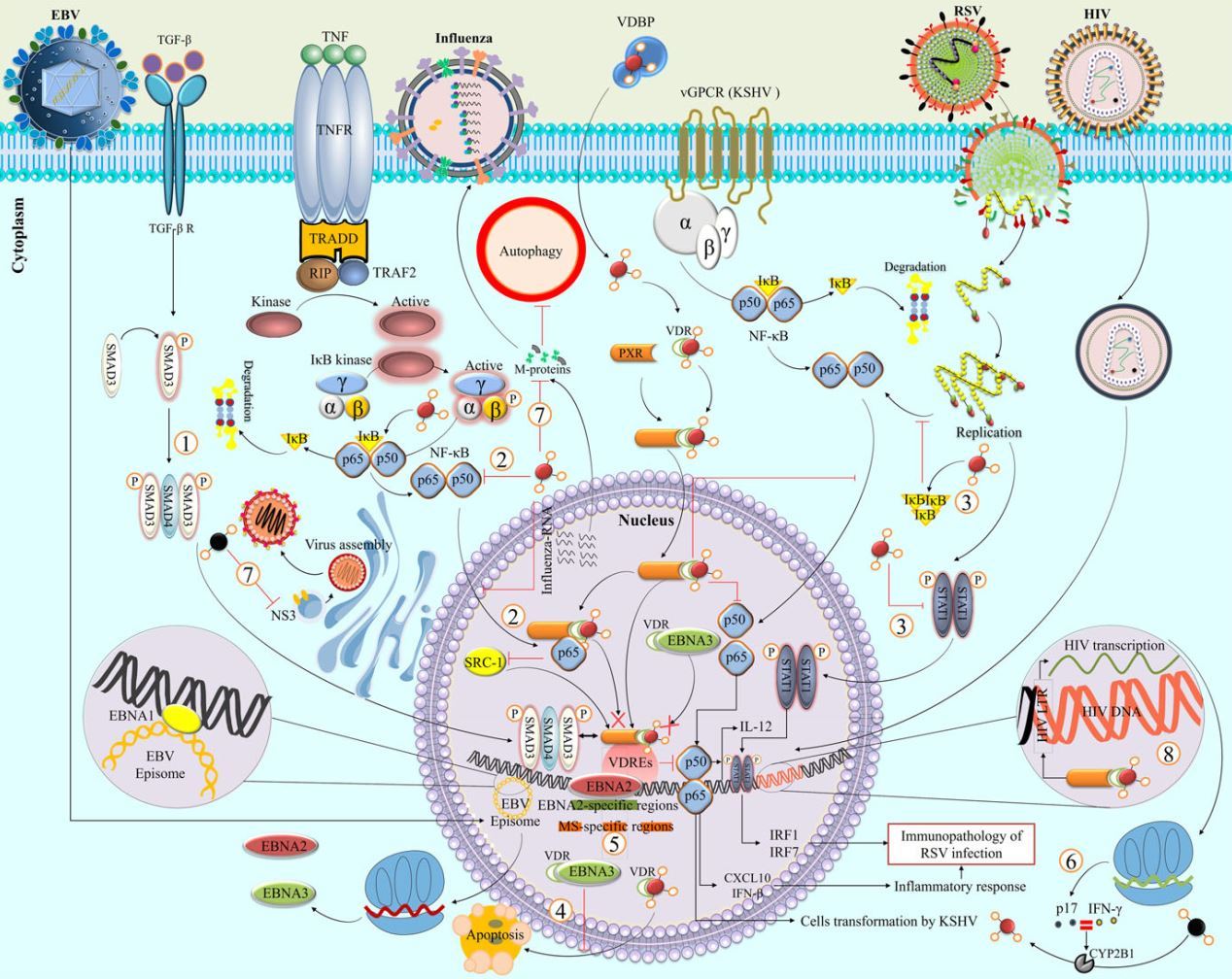
FIGURE 4 Vitamin D interaction with cellular and viral factors . Interaction between certain regulatory factors is a potential mechanism for the influence of vitamin D on virus infections such as (1) acting as a coactivator specific for ligand-induced transactivation of VDR that cross-talk between vitamin D and TGF-p signaling pathways (many viruses-Smad3), (2) inhibitory effect via integrating into the vitamin D-stimulated nuclear protein complex (many viruses-NF-KB subunits and TNF-a), (3) inhibition of vitamin D induced apoptosis (EBV-EBNA3), (4) overlapping with VDR binding sites (EBV-EBNA2), (5) vitamin D reduced immunopathology of viral infections, probably by inducing the inflammatory inhibitors (RSV- kBa) and preventing its translocation into the nucleus and subsequent binding to DNA promoter regions (RSV-NF-kB, IL-12,IRF1, IRF7, and pSTAT-1), (6) inducing CYP27B1 activity (HIV p17 matrix protein), (7) vitamin D decreased viral proteins (influenza M and HCV NS3 proteins), (8) and activated of viral replication (HIV-1 LTR). Abbreviation: Phosphorylated STAT-1 (pSTAT-1), retinoic X receptor (RXR), steroid receptor coactivator 1 (SRC-1), vitamin D receptor (VDR), viral G protein-coupled receptor associated to Kaposi sarcoma (vGPCR)
The pleiotropic role of vitamin D has been explored over the past decades and there is compelling evidence for an epidemiological association between poor vitamin D status and a variety of diseases. While the potential anti-viral effect of vitamin D has recently been described, the underlying mechanisms by which vitamin D deficiency could contribute to viral disease development remain poorly understood.
The possible interactions between viral infections and vitamin D appear to be more complex than previously thought.
Recent findings indicate a complex interplay between viral infections and vitamin D, including
the induction of anti-viral state,
functional immunoregulatory features,
interaction with cellular and viral factors,
induction of autophagy and apoptosis, and
genetic and epigenetic alterations.
While crosstalk between vitamin D and intracellular signalling pathways may provide an essential modulatory effect on viral gene transcription, the immunomodulatory effect of vitamin D on viral infections appears to be transient. The interplay between viral infections and vitamin D remains an intriguing concept, and the global imprint that vitamin D can have on the immune signature in the context of viral infections is an area of growing interest.
