57 studies of factors that increase the risk of Autism (Vitamin D DECREASES the risk)
Determinants of Autism Spectrum Disorder
J Indep Med 2026 Vol. 2 No. 3 50 pagesNicolas Hulscher, John S. Leake1, Simon Troupe1, Claire Rogers1, Kirstin Cosgrove1, M. Nathaniel Mead1, Breanne Craven1, Mila Radetich1, Andrew Wakefield, Peter A. McCullough1
57 minute inerview of 2 of the authors
Background: Autism spectrum disorder (ASD) now affects more than one in 31 children in the United States, with prevalence rising sharply over recent decades. ASD is recognized as a complex neurodevelopmental disorder shaped by genetic, environmental, and iatrogenic influences. Clarifying the contribution of these determinants is critical to addressing the escalating public health burden.
Methods: We comprehensively examined epidemiologic, clinical, and mechanistic studies evaluating potential ASD risk factors, assessing outcomes, exposure quantification, strength and independence of associations, temporal relationships, internal and external validity, overall cohesiveness, and biological plausibility.
Results: Key determinants of new-onset ASD before age nine include advanced parental age, premature delivery, genetic variants, sibling recurrence, maternal immune activation, in utero drug exposure, environmental toxicants, gut-brain axis disruption, and cumulative routine childhood vaccination. These factors may converge through pathways such as immune dysregulation, mitochondrial dysfunction, and neuroinflammation, which may contribute to neurodevelopmental injury in susceptible children. Of 136 studies examining childhood vaccines or their excipients, 29 found neutral risks or no association, while 107 inferred a possible link between immunization or vaccine components and ASD or other neurodevelopmental disorders (NDDs), based on findings spanning epidemiologic, clinical, mechanistic, neuropathologic, and case-report evidence of developmental regression.
12 studies comparing fully vaccinated and completely unvaccinated populations consistently showed superior overall health outcomes among the unvaccinated, including significantly lower risks of chronic disease and neuropsychiatric disorders such as ASD. The neutral association papers were undermined by absence of a genuinely unvaccinated control group, registry misclassification, ecological confounding, and averaged estimates that obscure effects within vulnerable subgroups. We observed strong, consistent increases in cumulative vaccine exposure during early childhood and the reported prevalence of autism across successive birth cohorts. To date, no study has evaluated the safety of the entire cumulative pediatric vaccine schedule for neurodevelopmental outcomes through age 9 or 18 years.
Conclusion: The totality of evidence supports a multifactorial model of ASD in which genetic predisposition, neuroimmune biology, environmental toxicants, perinatal stressors, and iatrogenic exposures converge to produce the phenotype of a postencephalitic state. Combination and early-timed routine childhood vaccination represents a significant modifiable risk factor for ASD within a broader multifactorial framework, supported by convergent mechanistic, clinical, and epidemiologic findings, and characterized by intensified use, the clustering of multiple doses during critical neurodevelopmen- tal windows, and the lack of research on the cumulative safety of the full pediatric schedule. As ASD prevalence continues to rise at an unprecedented pace, clarifying the risks associated with cumulative vaccine dosing and timing remains an urgent public health priority.
Claude AI detailed summary of study
Parental & Genetic Factors
| Factor | Risk Estimate | Source (study cited) |
|---|---|---|
| Paternal age >40 vs <30 | 5.75× | Sandin et al. |
| Paternal age >50 | ~8× mutation transmission vs age 20 | Sandin et al. |
| Older mother (advanced age) | aOR 1.47 (1.33–1.62) | Dehesh et al. |
| Older father | aOR 1.51 (1.40–1.62) | Dehesh et al. |
| Mother >40 vs 25–29 | +40–80% | meta-analytic |
| MTHFR C677T (dominant model) | OR 1.47 (1.13–2.93) | Razi et al. |
| Sibling with ASD (recurrence) | 5–10× general population | Hansen; Ozonoff (20.2% by age 3) |
Maternal/Perinatal Factors
| Factor | Risk Estimate | Source |
|---|---|---|
| Preterm birth (<37 wk) | OR 3.3 | Laverty et al. |
| Maternal influenza vax in pregnancy | Crude HR 1.22 (1.14–1.31); adj. 0.97 | Neeman 2025 |
| 1st-trimester influenza vax | aHR 1.20 (1.04–1.39) | Zerbo 2017 |
| RSVpreF vaccine → preterm delivery | RR 1.24 (1.08–1.44) | Marchand meta-analysis |
| Elevated maternal CRP | + association | Brown et al. |
| Chorioamnionitis | + association | Tsamantioti |
| Maternal autoimmune disease | + association | Chen meta-analysis |
| Maternal diabetes (GDM/T1/T2) | + association | Xiang et al. |
| Maternal obesity | + association | Li, Krakowiak |
| Hypertensive disorders of pregnancy | + association | Maher, Brand |
| Maternal hypothyroxinemia | + association | Román |
| Gestational vitamin D deficiency | + autism-related traits | Vinkhuyzen |
| Neonatal jaundice | + association | Kujabi, Jenabi |
| Assisted reproductive technology | + association | Djuwantono, Andreadou |
| Maternal anemia | + association | Wiegersma |
Environmental Toxicants
| Factor | Risk Estimate | Source |
|---|---|---|
| Maternal pesticide exposure (pooled) | OR 1.19 (1.04–1.36) | Xu meta-analysis |
| Regional pesticide use (case-control) | OR 1.34 (1.24–1.44) | Roman |
| Heavy metals (Cd, Pb, As, Hg) in hair/blood/urine | Elevated vs controls | Ding (53 studies) |
| Traffic-related air pollution / PM | + association | Volk, Pagalan |
| Prenatal NO₂ | + association | Murphy |
| Industrial As/Pb/Hg facility proximity | + association | Dickerson |
| Prenatal PFAS | + association | Ames |
| Gestational phthalates | + autistic traits | Oulhote/Lanphear |
| Prenatal PBDE flame retardants | modest + | Hertz-Picciotto |
| Lithium in drinking water | + association | Liew |
In Utero Pharmacological Exposures
| Drug | Risk Estimate | Source |
|---|---|---|
| Valproate | 2.9× ASD; 5.2× childhood autism | Christensen |
| Other antiseizure meds | drug-specific gradients | Bjørk |
| SSRIs (2nd/3rd trimester) | +87% | Boukhris (disputed; Ames null) |
| Antipsychotics | RR 1.10 (0.98–1.24) ASD; mostly attenuated | Wang meta-analysis |
| Opioids | + association (heterogeneous) | Balalian |
| Acetaminophen (medium/high dose) | <20% excess, attenuated to null in sibling controls | Ahlqvist |
| Acetaminophen + post-MMR | OR 6.11 (1.42–26.3) | Schultz |
| Prenatal antibiotics | aHR 1.10 (1.01–1.19), null in sibling models | Hamad, Choi |
Childhood Vaccines (positive-association studies)
| Exposure | Risk Estimate | Source |
|---|---|---|
| Thimerosal-containing DTaP vs free | Autism OR 1.8; speech OR 2.1; thinking abn. OR 8.2 | Geier 2004 |
| Per 25 µg ethyl-Hg from Hib | Autism OR 1.49; tics 1.43; ADHD 1.50 | Geier 2017 |
| Verstraeten Phase 1 (>25 µg Hg in 1st month) | NDD RR 1.8 (1.1–2.8); autism RR 7.6 (1.8–31.5) | Verstraeten EIS 1999 |
| Hepatitis B triple series (boys, EIS) | OR 8.63 (2.08–35.8) | Gallagher 2008 |
| Neonatal HepB (male) | Autism OR 3.00 (1.11–8.13) | Gallagher & Goodman 2010 |
| 3 thimerosal HepB doses (37.5 µg Hg) | Developmental delay OR 3.07 | Geier 2014 |
| MMR <36 mo, African-American boys | OR 2.25 (1.25–4.03) | Hooker reanalysis |
| Aluminum per 1 mg by age 2 (Asperger's) | aHR 1.67 (1.01–2.77) | Jablonowski & Hooker |
| MMRV vs MMR+V (febrile seizure) | ~2× | Klein |
| MMR febrile seizure (children with personal seizure hx) | 19.47/1,000 doses | Vestergaard |
Vaccinated vs. Completely Unvaccinated Cohorts
| Study | ASD/NDD Risk Estimate |
|---|---|
| Mawson 2025 (Florida Medicaid, n=47,155) | ASD RR 4.4 (2.85–6.84) at ≥11 vaccination visits; preterm+vax NDD aOR 3.58 |
| Lamerato/Henry Ford (n=18,468) | NDD aHR 5.53 (2.91–10.51); ASD 23 vax vs 1 unvax; any chronic condition aHR 2.54 |
| Hooker & Miller 2021 (n=1,565) | Autism OR 5.03 (1.64–15.5); ADHD OR 20.8 (4.74–91.2); GI OR 13.8; asthma OR 17.6 |
| Mawson 2017 (homeschool, n=666) | Preterm+vax vs term+unvax NDD OR 14.5 |
| Hooker & Miller 2020 (n=4,821) | Developmental delay OR 2.18 (1.47–3.24); asthma OR 4.49; ear infection OR 2.13 |
| Lee/Amish 2010 | 1 in 270 ASD vs CDC 1 in 68 (same era) |
| Gallagher 2008 (NHANES, HepB boys) | EIS OR 8.63 (2.08–35.8) |
Other
- Gut–brain axis/dysbiosis: ASD children ≥4× more GI problems; mechanism via Clostridia overgrowth, intestinal permeability, microbial metabolites.
- Febrile seizures: 41% rate of NDD by age 9–10 in children with febrile seizures (Nilsson); strong association with ASD, DCD, intellectual disability (Gillberg).
- Mitochondrial dysfunction / oxidative stress: mechanistic, no direct OR — proposed amplifier of all exposure effects.
Caveats Henry should note about this source:
- Authors are McCullough Foundation + Andrew Wakefield (Wakefield Media Group); this is an advocacy-leaning narrative review, not a systematic review with formal risk-of-bias scoring.
- Many of the largest vaccine-positive estimates (Mawson, Hooker, Garner, Lamerato Senate-submitted data) come from surveys, claims data without clinical verification, or unpublished/legal-record sources — the authors themselves acknowledge this limitation.
- Mainstream meta-analyses (Taylor 2014, Hviid 2019, Madsen 2002, Jain 2015, DeStefano 2013, Andersson 2025) — included in this paper's "neutral" 29 — find null associations for MMR, thimerosal, and aluminum, though this paper argues they suffer from misclassification and lack of true unvaccinated controls.
- The Andersson 2025 erratum / Jablonowski-Hooker reanalysis is a live methodological dispute, not a settled finding.
Claude AI; High vitamin D during pregnancy weeks 14-17 reduces the risk of Autism
The research consistently points to mid-gestation (the second trimester) as the most critical window, though adequate vitamin D throughout pregnancy matters.
The second trimester (roughly weeks 14–27) appears to be the most important period. Studies from Australia and the Netherlands found that mothers with vitamin D deficiency specifically during mid-gestation were more than twice as likely to give birth to an autistic child. A large study of 4,334 pregnant women confirmed this association across different ethnic backgrounds. Some research finds that deficiency during the second trimester has a more pronounced effect on autism risk than at other stages. doublecareaba
Why This Period Matters
The second trimester is a period of intense fetal brain development — vitamin D regulates gene expression, cellular differentiation, calcium signaling, and neurotrophic factors all critical to proper brain formation. When vitamin D is deficient during this window, animal studies show it can cause overproliferation of neuronal cells and elevated testosterone in the fetal brain, both linked to autism-like behaviors. Fetal brains depend entirely on maternal vitamin D, since the circulating form crosses the placental barrier. advancedtherapyclinic
Risk Magnitude and Threshold
Children born to mothers deficient during mid-pregnancy had up to a 4-fold higher likelihood of ASD omegor
Deficiency defined as serum levels below 25–30 nmol/L (10–12 ng/mL) carries the highest risk advancedtherapyclinic
- Neonatal vitamin D levels at birth also predict ASD risk — low levels at birth correlate with higher ASD prevalence advancedtherapyclinic
Early Pregnancy and Preconception Also Count
Higher maternal vitamin D levels before the intervention (preconception/early pregnancy) were also associated with decreased autism risk, lower autistic symptom load, and reduced ADHD risk. This suggests that building adequate stores before and throughout pregnancy — not just mid-gestation — is the safest approach, similar to how folic acid must be present early to prevent neural tube defects. sciencedirect
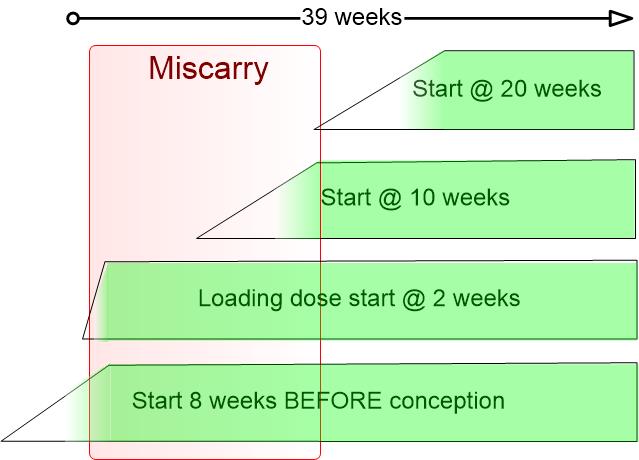
Need a Vitamin D loading dose by week 10 to reduce Autism risk (if not already started)
Ensure a healthy pregnancy and baby - take Vitamin D before conception has:
Related in Vitamin D Life
- Increase in Autism prevalence matches increase in Vaccinations
- 17 Autism risk factors: low Vitamin D, virus, vaccine, mercury etc. - many studies
- 20+ chronic stressors result in poor health about 80% overlap with Autism stressors
- Overview Autism and vitamin D
- Autism associated with low Vitamin D - PhD dissertation - Feb 2025
- Autism IS associated with childhood vaccines - CDC reversal Dec 2025
Autism rate in siblings reduced 4X by vitamin D: 5,000 IU during pregnancy, 1,000 IU to infants
Autism treated by Vitamin D: Dr. Cannell - video has the following reasons:
Both have strong inheritance features – Vitamin D about 60%
Both have gotten substantially worse in last 30 years
Vitamin D is known to be involved in brain development
All autistic children are VitD deficient, but not all children who are deficient are autistic: genes are involved
When giving vitamin D to cure children of rickets, “mental dullness” decreases as well
Children with genes which give them too much (Williams Syndrome) have to reverse of autism – too sociable
Mothers having lots of fish (and thus more vitamin D) give birth to kids with less autistic symptoms
Both are associated with weak bones
Both worse around the age of weaning
Autism is more common in rich families – more likely to apply sun screen and stay indoors
Autism increases with drugs which lower levels of vitamin D
Seizures are common with Autism - Vitamin D has been shown to reduce seizures
Fewer autistic symptoms (such as sleep problems) during summer: when child gets more vitamin D from the sun
Both worse with latitude
Both vary with Ultraviolet light
Both vary with time of year (more birth of autistics in March in Northern hemisphere)
2X more urban autism – less UVB in urban environments
Both worse with pollution
Both worse with increased clouds and rain
Both are worse with closely spaced pregnancies
Autistics have an abnormal immune response – similar to that of vitamin D deficiency
Low levels of vitamin D in mother animals reduce brain function in offspring
Vitamin-deficient rat pups have similar brain abnormalities to those of human autistic children
Autistic children get less vitamin D in their blood for the same amount of sun exposure
The 4 males/1 female ratio - Note estrogen increases vitamin D in the brain (testosterone does not)
Both worse in African Americans (A-A 2-3 increased autism rate)
Both worse in Dark-skinned immigrants in Europe