Severe Sepsis associated with very low vitamin D
Vitamin D Deficiency in Human and Murine Sepsis.
Parekh, Dhruv MRCP; Patel, Jaimin M. FRCA; Scott, Aaron PhD; Lax, Sian PhD; Dancer, Rachel C. A. MRCP; D'Souza, Vijay PhD; Greenwood, Hannah PhD; Fraser, William D. MD; Gao, Fang MD; Sapey, Elizabeth PhD; Perkins, Gavin D. MD; Thickett, David R. DM
Critical Care Medicine. AN: 00003246-900000000-96779. Ahead of Print, 14 Sept 2016
Objectives: Vitamin D deficiency has been implicated as a pathogenic factor in sepsis and ICU mortality but causality of these associations has not been demonstrated. To determine whether sepsis and severe sepsis are associated with vitamin D deficiency and to determine whether vitamin D deficiency influences the severity of sepsis.
Design, Setting, and Patients: Sixty-one patients with sepsis and severe sepsis from two large U.K. hospitals and 20 healthy controls were recruited. Murine models of cecal ligation and puncture and intratracheal lipopolysaccharide were undertaken in normal and vitamin D deficient mice to address the issue of causality.
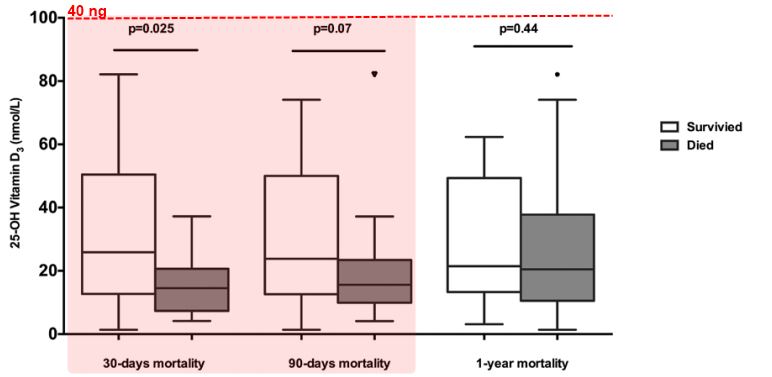
Measurements and Main Results: Patients with severe sepsis had significantly lower concentrations of 25-hydroxyvitamin D3 than patients with either mild sepsis or age-matched healthy controls (15.7 vs 49.5 vs 66.5 nmol/L; p = 0.0001). 25-hydroxyvitamin D3 concentrations were significantly lower in patients who had positive microbiologic culture than those who were culture negative (p = 0.0023) as well as those who died within 30 days of hospital admission (p = 0.025).
Vitamin D deficiency in murine sepsi s was associated with increased peritoneal (p = 0.037), systemic (p = 0.019), and bronchoalveolar lavage (p = 0.011) quantitative bacterial culture. This was associated with reduced local expression of the cathelicidin-related antimicrobial peptide as well as evidence of defective macrophage phagocytosis (p = 0.029). In the intratracheal lipopolysaccharide model, 1,500 IU of intraperitoneal cholecalciferol treatment 6 hours postinjury reduced alveolar inflammation, cellular damage, and hypoxia.
Conclusions: Vitamin D deficiency is common in severe sepsis. This appears to contribute to the development of the condition in clinically relevant murine models and approaches to correct vitamin D deficiency in patients with sepsis should be developed.
Clipped from PDF - with bolding and bullet points added by Vitamin D Life
DISCUSSION
We have confirmed, in a cohort of hospitalized patients with sepsis, that VDD is common, severe, and is associated with disease severity, bacterial positive culture, and 30-day mortality. To demonstrate causation of VDD as a driver of sepsis severity, our CLP mouse studies demonstrated exaggerated bacterial growth both locally and systemically, increased cellular inflammation, and dysregulated accumulation of apoptotic neutrophils in VDD mice. Using the IT-LPS challenge model, we demonstrate that the novel administration of IP cholecalciferol is an effective postinjury therapy when given 6 hours postinjury.
We enrolled a mixed population of both mild and severe sepsis patients. VDD was both common and severe in patients with severe sepsis. 25(OH)D3 concentrations were lower in patients who died than survived as well as patients who grew culture positive bacterial specimens. Additionally, clinical markers of sepsis severity (lactate, metabolic acidosis) were associated with lower levels of 25(OH)D3 suggesting perhaps these measures could reflect a VDD population in sepsis. A criticism often levelled at observational studies, such as ours, is whether the VDD is a marker of critical illness or a mechanistic driver. Two recent meta-analyses of observational studies have confirmed a significant association between vitamin D status and susceptibility to sepsis (18), rates of infection, and 30-day mortality (17). Our findings are concordant with observational studies that have demonstrated that low vitamin D status upon admission is associated with sepsis (16), bacteremia (25), and acute respiratory distress syndrome (26, 27).
The murine studies sought to establish whether inducing VDD by diet before injury in mice leads to exaggerated sepsis and enhanced cellular inflammation/ dysfunction. We successfully established severe deficiency in the mice, with concentrations of 25(OH)D3 similar to those who died from sepsis in our clinical cohort. This deficiency was reflected also in reduced circulating 1,25(OH)2D3, the bioactive form of vitamin D. In contrast, our WT mice had 25(OH)D3 and 1,25 (OH)2D3 concentrations similar to our mild sepsis patient population.
In the clinically relevant CLP model of early sepsis, VDD was associated with
exaggerated bacterial growth in the peritoneal cavity,
elevated systemic bacteremia as well as
increased bacterial translocation to the alveolar compartment.
This was associated with abnormal protein permeability of the peritoneal and alveolar capillary barrier. In the PLF, there was exaggerated cellular inflammation in VDD mice with evidence of impaired antibacterial responses in terms of CRAMP release and the ability of peritoneal macrophages to phagocytose E. coli. These cellular changes resulted in increased accumulation of apoptotic neutrophils in the PLF. Previous animal studies have shown a benefit of 1,25(OH)2D3 on sepsis-induced coagulopathy in rats (28) and our CRAMP results confirm findings by others of decreased antimicrobial peptide in VDD in sepsis and critical illness (29, 30). Our study is the first to report VDD as a predeterminant of sepsis and decreased macrophage phagocytosis in a relevant murine model. These data support our hypothesis that VDD is mechanistically important in driving sepsis and led us to the question of whether treating deficiency postinjury would be an effective therapy.
In the United Kingdom, the regulatory framework for animal experiments dictated that we could not keep VDD animals alive post-CLP for more than 16 hours because of serious adverse events so we were only able to model early sepsis using this technique. Our group has recently shown a detrimental effect of VDD with exaggerated lung injury, dysregulated cellular inflammation, and apoptosis, which manifested as reduced oxygenation in an IT-LPS direct murine lung injury model 48 hours after injury (19). We, therefore, used our IT-LPS model to test whether postinjury treatment of VDD mice attenuated the effects of VDD upon inflammatory injury.
Traditionally, vitamin D supplements have been given
by mouth,
intramuscular injection (cholecalciferol and ergocalciferol), or
by IV infusion (calcitriol)
with mixed results potentially due to
poor absorption from muscle
or the gut
or a short IV half-life (31-33).
We elected to test the effect of IP administration of **1,500 IU cholecalciferol liquid as a novel route to restore VDS—a dose that proved effective in restoring 25(OH)D3 concentrations back to those seen in WT mice. Postinjury cholecalciferol therapy was effective in reducing the
exaggerated cellular inflammation,
alveolar epithelial damage as measured by PPI and RAGE release, and reduced
hypoxia (oxygen saturations)
when given 6 hours after the insult supporting IP administration of cholecalciferol as a novel potential route of administration in patients as well as evidence that restoration of vitamin D levels may reduce inflammation with physiologic benefit.
This study has limitations. First, patients were recruited up to 48 hours after hospital admission, so it is possible that the 25(OH)D3 concentrations seen reflected changes associated with sepsis rather than the cause. Second, this is a retrospective study of a small number of patients that could not control for patient comorbidities. The effects of sepsis and critical illness on the vitamin D metabolome are unknown and this complex interplay needs prospective large-scale studies that consider other preinsult comorbidities, chronic illness, nutritional status, and other confounders. It was for this reason we did the murine studies. In our CLP model, we studied early sepsis due to restrictions from the animal ethics committee. This meant that our animals had limited alveolar damage, which was why we undertook additional studies in the IT-LPS model. Although the VDD induced by diet design investigated whether pre-existing VDD is causal and a mechanistic driver to the severity of sepsis rather than the consequence of the sepsis insult in the murine model, it may not wholly explain the findings of the human study due to the lack of vitamin D status before sepsis and its effects on vitamin D status as discussed above. Finally, we have shown the effects of VDD in only two models of murine lung injury. Further work in other models related to sepsis particularly experimental pneumonia need to be undertaken.* In conclusion, we suggest that therapies aimed at restoring VDS in patients at risk of deficiency when they are admitted to hospital need to be developed to try and prevent the increasing healthcare burden of sepsis patients. Key to this will be establishing appropriate dosing regimens for vitamin D replacement in the critically ill patients both within and outside the ICU. *#### REFERENCES
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 1992; 20:864-874
Dellinger RP, Carlet JM, Masur H, et al; Surviving Sepsis Campaign Management Guidelines Committee: Surviving Sepsis Campaign guidelines for management of severe sepsis and septic shock. Crit Care Med 2004; 32:858-873
Nathani N, Perkins GD, Tunnicliffe W, et al: Kerbs von Lungren 6 antigen is a marker of alveolar inflammation but not of infection in patients with acute respiratory distress syndrome. Crit Care 2008; 12:R12
Greenwood H, Patel J, Mahida R, et al: Simvastatin to modify neutrophil function in older patients with septic pneumonia (SNOOPI): Study protocol for a randomised placebo-controlled trial. Trials 2014; 15:332
van Schoor NM, Lips P: Worldwide vitamin D status. Best Pract Res Clin Endocrinol Metab 2011; 25:671-680
Shoben AB, Kestenbaum B, Levin G, et al: Seasonal variation in 25-hydroxyvitamin D concentrations in the cardiovascular health study. Am J Epidemiol 2011; 174:1363-1372
Sherman SS, Hollis BW, Tobin JD: Vitamin D status and related parameters in a healthy population: The effects of age, sex, and season. J Clin Endocrinol Metab 1990; 71:405-413
Hypponen E, Power C: Hypovitaminosis D in British adults at age 45 y: Nationwide cohort study of dietary and lifestyle predictors. Am J Clin Nutr 2007; 85:860-868
Danai PA, Sinha S, Moss M, et al: Seasonal variation in the epidemiology of sepsis. Crit Care Med 2007; 35:410-415
Parekh D, Thickett DR, Turner AM: Vitamin D deficiency and acute lung injury. Inflamm Allergy Drug Targets 2013; 12:253-261
Gombart AF, Borregaard N, Koeffler HP: Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihy- droxyvitamin D3. FASEB J 2005; 19:1067-1077
Cannell JJ, Hollis BW: Use of vitamin D in clinical practice. Altern Med Rev 2008; 13:6-20
Remmelts HH, van de Garde EM, Meijvis SC, et al: Addition of vitamin D status to prognostic scores improves the prediction of outcome in community-acquired pneumonia. Clin Infect Dis 2012; 55:1488-1494
Braun AB, Litonjua AA, Moromizato T, et al: Association of low serum 25-hydroxyvitamin D levels and acute kidney injury in the critically ill. Crit Care Med 2012; 40:3170-3179
Braun AB, Gibbons FK, Litonjua AA, et al: Low serum 25-hydroxyvitamin D at critical care initiation is associated with increased mortality. Crit Care Med 2012; 40:63-72
Moromizato T, Litonjua AA, Braun AB, et al:* Association of low serum 25-hydroxyvitamin D levels and sepsis in the critically ill. *Crit Care Med 2014; 42:97-107
de Haan K, Groeneveld AB, de Geus HR, et al:* Vitamin D deficiency as a risk factor for infection, sepsis and mortality in the critically ill: Systematic review and meta-analysis *. Crit Care 2014; 18:660
Upala S, Sanguankeo A, Permpalung N:* Significant association between vitamin D deficiency and sepsis: A systematic review and meta-analysis. *BMC Anesthesiol 2015; 15:84
Dancer RC, Parekh D, Lax S, et al: Vitamin D deficiency contributes directly to the* acute respiratory distress syndrome *(ARDS). Thorax 2015; 70:617-624
Patel JM, Snaith C, Thickett DR, et al: Randomized double-blind placebo-controlled trial of 40 mg/day of atorvastatin in reducing the severity of sepsis in ward patients (ASEPSIS Trial). Crit Care 2012; 16:R231
Owens DJ, Webber D, Impey SG, et al: Vitamin D supplementation does not improve human skeletal muscle contractile properties in insufficient young males. Eur J Appl Physiol 2014; 114:1309-1320
Marcinowska-Suchowierska E, Walicka M, Tatataj M, et al: Vitamin D supplementation in adults - guidelines. Endokrynol Pol 2010; 61:723-729
Lax S, Wilson MR, Takata M, et al: Using a non-invasive assessment of lung injury in a murine model of acute lung injury. BMJ Open Respir Res 2014; 1:e000014
Kovach MA, Ballinger MN, Newstead MW, et al: Cathelicidin-related antimicrobial peptide is required for effective lung mucosal immunity in Gram-negative bacterial pneumonia. J Immunol 2012; 189:304311
Quraishi SA, Litonjua AA, Moromizato T, et al: Association between prehospital vitamin D status and hospital-acquired bloodstream infections. Am J Clin Nutr 2013; 98:952-959
Thickett DR, Moromizato T, Litonjua AA, et al:* Association between prehospital vitamin D status and incident acute respiratory failure in critically ill patients: A retrospective cohort study *. BMJ Open Respir Res 2015; 2:e000074
Parekh D, Dancer RC, Lax S, et al: Vitamin D to prevent acute lung injury following oesophagectomy (VINDALOO): Study protocol for a randomised placebo controlled trial. Trials 2013; 14:100
M0ller S, Laigaard F, Olgaard K, et al:* Effect of 1,25-dihydroxy-vitamin D3 in experimental sepsis *. Int J Med Sci 2007; 4:190-195
Jeng L, Yamshchikov AV, Judd SE, et al: Alterations in vitamin D status and anti-microbial peptide levels in patients in the intensive care unit with sepsis. J Transl Med 2009; 7:28
Leaf DE, Croy HE, Abrahams SJ, et al:* Cathelicidin antimicrobial protein, vitamin D, and risk of death in critically ill patients. *Crit Care 2015; 19:80
Leaf DE, Raed A, Donnino MW, et al:* Randomized controlled trial of calcitriol in severe sepsis *. Am J Respir Crit Care Med 2014; 190:533-541
Amrein K, Schnedl C, Holl A, et al:* Effect of high-dose vitamin D3 on hospital length of stay in critically ill patients with vitamin D deficiency: The VlTdAL-ICU randomized clinical trial *. JAMA 2014; 312:1520-1530
Nair P, Venkatesh B, Lee P, et al:** A randomized study of a single dose of intramuscular cholecalciferol in critically ill adults . Crit Care Med 2015; 43:2313-2320 📄 Download the PDF from Vitamin D Life__
