Global Push for RCTs in COVID-19
Huge Global Push for RCTs in COVID-19: From Random to Randomized
Helen Leask; April 04, 2020 Medscape
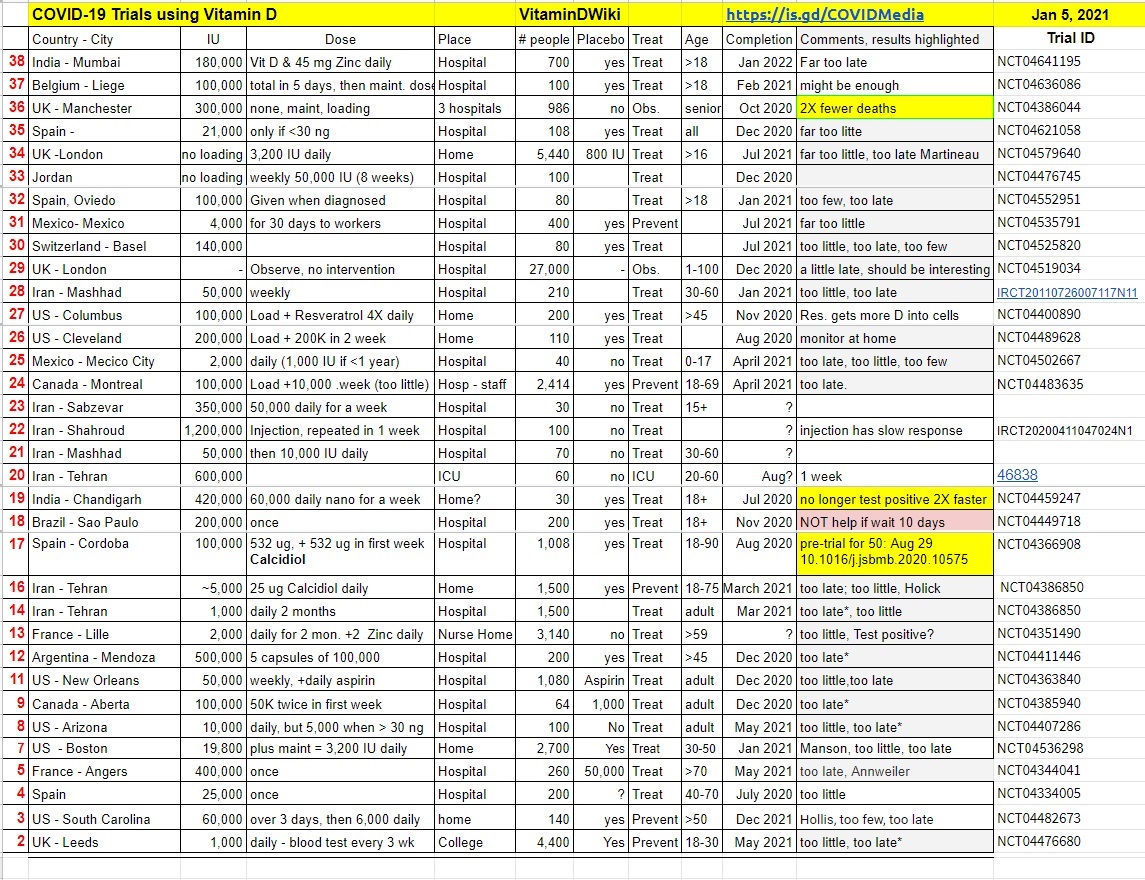
Updated list of trials in Vitamin D Life
📄 Download the Medscape PDF from Vitamin D Life
Editor's note: Find the latest COVID-19 news and guidance in Medscape'sCoronavirus Resource Center.Scientists and clinicians across the globe have responded to the ongoing coronavirus pandemic with a huge, high-quality global research effort to find a treatment for COVID-19.As shown in the Table (see below) of all COVID-19 randomized controlled treatment trials, almost 60 of such trials are planned, recruiting, or underway.This impressive roll-call of studies just 3 to 4 months after the COVID-19 outbreak is testament to the progress that has been made since theEbola outbreak in 2014, said Andre Kalil, MD, MPH, of the University of Nebraska Medical Center in Omaha.Kalil is leading the first clinical trial in the United States of an experimental treatment for COVID-19, the National Institutes of Health (NIH)-sponsored Adaptive COVID-19 Treatment Trial (ACTT)."Years ago it could take months or years to design a randomized controlled trial," Kalil told Medscape Medical News. "We learned during the Ebola outbreak that we've got to do much better than that. With the COVID-19 outbreak, we were able to get a randomized trial up and running in a matter of weeks. It's a very optimistic message: we can do that really fast, efficiently, and safely."Kalil emphasized the need for randomized controlled trials in a JAMAViewpoint article published last month."Without a control group, we can never, ever safely find new medications for any disease," Kalil commented.The problem with off-label use and compassionate use of drugs is interpretation of results. If the patient died, it's assumed he or she died of the disease, but if the patient survived, it's assumed he or she survived because of the given drug, but "this is not true," he said.Kalil emphasized there is no way to know whether patients actually benefit from a drug or are harmed by it without a randomized clinical trial 一 a well-established principle of evidence-based medicine that can be forgotten during a crisis.This tragedy of not discovering new therapies during an outbreak cannot be repeated. Dr Andre Kalil
Kalil cited the lesson of the 2014 Ebola outbreak, in which 30,000 individuals were infected and numerous therapies were tested, but no new drugs emerged, in part because virtually all studies were uncontrolled, single-arm endeavors. "This tragedy of not discovering new therapies during an outbreak cannot be repeated," he commented.Canadian researchers drew similar conclusions from the extensive use of oseltamivir during the 2009H1N1 pandemic. In acommentary in the Canadian Medical Association Journal, Matthew Cheng, MDCM, of McGill University Health Centre in Montreal, Canada, and colleagues wrote, "Even now, because there has been no RCT, it is not definitely known whether oseltamivir is efficacious…"The Canadian authors conclude, "There is a strong ethical and clinical argument for replacing such 'random' care with randomized care."Many of the larger studies are collaborative, multinational trials linking study initiators in the United States, Canada, China, and France with clinical researchers in Southeast Asia, Australasia, and a dozen European countries.China is currently running more randomized treatment trials than any other country, closely followed by the United States. So far, most countries hit hard by COVID-19 are hosting at least one randomized study, with the exception of Spain and Iran. This will likely change over time — the World Health Organization (WHO) is working on arandomized, multicenter adaptive trial that will cover "multiple sites," so far unspecified.WHO has identified a list of "promising candidates" for COVID-19 treatment. These include remdesivir (an investigational agent); lopinavir-ritonavir (approved for use in HIV) with or without interferon; investigational immunotherapies such as monoclonal and polyclonal antibodies; and convalescent sera.In its January 27 statement, WHO did not support the antimalarial chloroquine (or hydroxychloroquine),ribavirin (used for hepatitis), or corticosteroids/steroids for COVID-19 clinical studies.WHO is encouraging adaptive trial designs that test candidate drugs in sequence and can be launched quickly.[]Four Multinational Trials Underway]Four COVID-19 multinational adaptive trials are already underway, one starting with the investigational agent remdesivir, and two with theHIV drug combination of lopinavir-ritonavir. The fourth has four therapy groups from the outset: remdesivir, lopinavir-ritonavir with or without interferon, and hydroxychloroquine.Remdesivir is the first agent investigated in the NIH-sponsored trial ACTT, headed by Kalil at the University of Nebraska.A broad-spectrum antiviral agent, remdesivir (GS-5734, Gilead Sciences Inc) has been studied as a potential treatment for Ebola, Marburg, MERS, and SARS without success. Kalil said remdesivir was chosen as the kick-off drug candidate for the NIH COVID-19 study on the basis of data from cell culture and two animal models.(Two other single-country trials are studying remdesivir, one in mild/moderate patients and one in severe COVID-19. Both are supported by the Chinese Academy of Medical Sciences. The NIH took into account the Chinese study designs in the creation of ACTT, as well as WHO's adaptive trial design.)The first results on remdesivir are expected soon. The two Chinese studies aim to report first results as early as April, and Kalil said results of the ACTT trial will be available "within a few weeks."Gilead Sciences, the developer of remdesivir, is in the process of making the drug available under expanded access instead of unwieldy, individual compassionate-use requests, to cope with "overwhelming demand." After a global outcry, it also recentlyrescinded its request for orphan drug designation for remdesivir, which would have granted the company market exclusivity for 7 years.The two other multinational adaptive studies are starting with lopinavir-ritonavir, a protease-inhibitor combination indicated for HIV/AIDS prevention and treatment. In combination with interferon B, it has shown effectiveness against another coronavirus, MERS, in animal models. However,results of lopinavir-ritonavir against COVID-19 have thus far been disappointing.In North America, the study of lopinavir-ritonavir is being spearheaded by Canada with two trials. The Canadian-driven adaptive CATCO trial is the largest treatment study in the country and is part of WHO's solidarity protocol for hospitalized patients. After lopinavir-ritonavir, the multinational team plans to look at hydroxychloroquine and remdesivir.The other Canadian adaptive trial of lopinavir-ritonavir is REMAP-CAP, a study ofcommunity-acquired pneumonia that was already running and has now folded in COVID-19 patients not in the CATCO study.The first results of these Canadian-run trials could be available as early as May.The fourth large trial is a European adaptive treatment trial. The DisCoVeRy trial has four parallel therapy groups and is testing lopinavir- ritonavir, either with or without interferon, against remdesivir and hydroxychloroquine.Other antivirals being tested against COVID-19 are arbidol,darunavir, favipiravir, oseltamivir, and various protease-inhibitor combinations in trials in China and Thailand. The WHO has just launched the SOLIDARITY trial, a randomized, multicenter, adaptive trial of antivirals currently involving 45 countries, and counting.Other therapies recommended by WHO for further investigation include monoclonal antibodies and other investigational immunotherapies. There has been intense interest in the use of interleukin-6 inhibitors, for example tocilizumab (Actemra, Genentech), for use in cytokine release storm in COVID-19 pneumonia, which is similar to use of these agents after CAR T-cell therapy. A total of 10 randomized clinical trials of these products against COVID-19 are currently running, spanning the United States, Canada, China, and Europe.Convalescent plasma is also recommended for further investigation by WHO, and studies are ongoing. The US Food and Drug Administration approvedemergency use of convalescent plasma for seriously ill COVID-19 patients on March 28.[]Hydroxychloroquine and Chloroquine] Despite enthusiastic endorsement by President Trump afterreports of positive results from an open-label French study, hydroxychloroquine and chloroquine are not currently recommended for further investigation in COVID-19 by WHO.Two COVID-19 randomized clinical trials with hydroxychloroquine have reported results, both from China. A study of 30 patients with early COVID-19 treated in the Shanghai Public Health Center,published in February, found that hydroxychloroquine 400 mg per day was no more effective than standard care. Using the same dose in a similar patient population, a non-peer-reviewed trial of 62 patients at the Renmin Hospital in Wuhan,posted on April 2, found that hydroxychloroquine reduced the time to clinical recovery.Five large hydroxychloroquine randomized controlled trials with sites in Canada, the United States, China, Europe, and Brazil — aiming for 8000 patients — are currently in the works.Despite the controversies, the overwhelming international collaborative effort in COVID-19 science is cause for hope, said Kalil, who has no financial conflicts of interest."My hope is that we're going to find a way to slow down and stop outbreaks for the future," Kalil said. If one looks at the many trials by researchers from multiple countries, "I think it shows that once all of us combine efforts, we can find solutions fast, save lives, and be better prepared for future outbreaks."[]Tracking the Trials]Several groups are tracking clinical trials of both therapeutics and vaccines for COVID-19, including the Milken Institute COVID-19 tracker, the Oxford Trials Tracker on COVID-19, and the Centre for Evidence-Based Medicine (CEBM) COVID-19 Registered Trials Tracker.Medscape has compiled a table (see below) of 50+ randomized clinical trials of treatments for COVID-19 that are currently ongoing and planned, with estimated completion dates. (Data sources includeclinicaltrials.gov, WHO R&D; Blueprint, and news feeds. Data are current as of April 3.)
